Screening Tests for Infections Transmissible by Transfusion
The organisms likely to be transmitted by transfusion are usually those, which are prevalent in a particular geographic area or population.
Organisms transmissible by transfusion are listed in Table 1197.1.
Prions
Bacteria
Parasites
|
According to studies, the prevalence of transmission of infections through transfusions is significantly higher in underdeveloping nations as compared to developed nations. Some of the reasons include:
- Proportion of replacement and professional donations is high Concealment of relevant medical history by prospective donors
- Specificity and sensitivity of screening tests for infections may be poor
- Non-implementation of National Transfusion Policy
- Repeat donor system is non-existent
- Collection of donor blood during window period In India, pre-transfusion testing of donor blood for agents listed in Table 1197.2 is currently mandatory.
| Disease | Tests |
| 1. Syphilis | Serologic test like Venereal Disease Research Laboratory (VDRL) test |
| 2. Hepatitis B | Hepatitis B surface antigen (HBsAg) |
| 3. Human immunodeficiency virus (HIV) infection | Anti-HIV 1 and anti-HIV 2 antibodies* |
| 4. Hepatitis C | Anti-HCV antibodies• |
| 5. Malaria | Blood smear |
| * Window periods for HIV and HCV infections are further reduced if tests for HIV RNA and HCV RNA (nucleic acid testing or NAT) are added. NAT, however, is not currently mandatory in India |
The importance of mandatory testing for infectious organisms transmissible by transfusion is: (i) some carriers of disease are often asymptomatic, (ii) some viral infections have long incubation periods, and (iii) it is essential to safeguard the health of the recipient.
Infections by hepatitis B and C viruses and HIV-1 and HIV-2 are characterized by:
- Persistence of organisms in circulation for prolonged duration without necessarily causing clinical manifestations
- Persistence in high titers
- Long incubation period
- Ability to cause chronic carrier state
- Viability of organisms in blood stored at 4°-6°C.
Following principal measures can prevent transmission of infection through transfusion:
- Blood should be collected only from voluntary, nonremunerated donors. All high risk persons (intravenous drug abusers, homosexuals, prostitutes, and sexual partners of such persons) and professional donors should be excluded. Standard criteria for selection of blood donors should be followed.
- All blood donations should be tested for infectious agents by screening tests; addition of further screening tests like HIV RNA and HCV RNA will further reduce the risk.
- Universal hepatitis B virus vaccination
- Observation of universal precautions in blood collection, processing, storage, and transfusion
- Leukofiltration of blood products
- Stringent quality control measures.
VIRUSES
Hepatitis B Virus (HBV)
HBV is a partially double-stranded DNA virus of 42 nm diameter (Figure 1197.1). It can cause acute hepatitis, chronic hepatitis, asymptomatic carrier state, cirrhosis, or hepatocellular carcinoma.
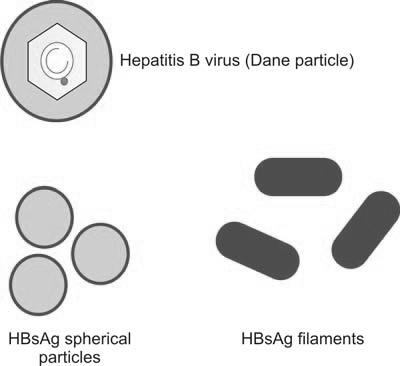
In India, prevalence of HBsAg carriers is reported to be 1.5 to 4%. HBV is highly infectious and is transmitted through all blood components and most of the blood derivatives. According to one study in India, incidence of post-transfusion hepatitis following a single transfusion was 6.7%; this was much higher in multi-transfused patients like patients with thalassemia and hemophilia. Infected hepatocytes release large amounts of hepatitis B surface antigen (HbsAg) into the bloodstream. Presence of HbsAg indicates active infection. The serological marker first to appear in HBV infection is HBsAg (as early as 5 days after infection).
Screening of all blood donations for HbsAg has greatly reduced the risk of transmission of HBV through transfusion.
Tests for screening donor blood for HbsAg are:
- Reverse passive hemagglutination assay (RPHA)
- Enzyme linked immunosorbent assay (ELISA)
- Radioimmunoassay (RIA).
Commercial test kits for detection of HbsAg are available and the exact test procedure is provided with each kit. General principles of these tests are outlined below.
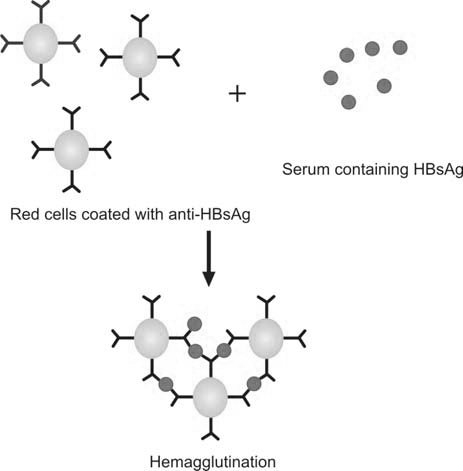
Reverse Passive Hemagglutination Assay
In RPHA, red cells that are coated with anti-HBs antibody are added to the donor’s serum. If HbsAg is present in the serum, agglutination of red cells will occur. Absence of agglutination indicates negative test. The test is called as ‘passive reverse agglutination’ because antibody, and not antigen, is artificially coated on to the red cells. The test is performed in U- or V-shaped wells of a microtiter plate which can be centrifuged to augment agglutination. The test is sensitive to 10-100 ng of HbsAg antigen per ml of serum (Figure 1197.2).
Enzyme-linked Immunosorbent Assay
Serum sample to be tested is incubated with anti-HbsAg antibody which has been coated to microtitre plate. The test serum is incubated in the well during which binding of antigen from serum (if present) and antibody (attached to the well) occurs. An enzyme-linked anti-HBs antibody (conjugate) is added which will combine with the bound antigen. A chromogenic enzyme substrate is finally added and the mixture is incubated in the dark. If enzyme is present (bound to the antigen), then its action on the substrate will lead to the colour development. A stopping solution (an acid) is added to prevent any further reaction between the enzyme and the substrate. The result is read in a spectrophotometer at the specified wavelength.
Hepatitis C Virus (HCV)
HCV is a single-stranded RNA virus of flaviviridae family. Incubation period is about 8 weeks. Most cases of HCV infection are asymptomatic. Some patients develop chronic hepatitis, cirrhosis, or hepatocellular carcinoma.
In India, prevalence of HCV is reported to be 1.66%. As the amount of HCV antigen in bloodstream is small and cannot be detected readily, screening of donor blood for HCV infection is done by detection of anti-HCV antibody in serum (becomes detectable after 6-8 weeks of infection).
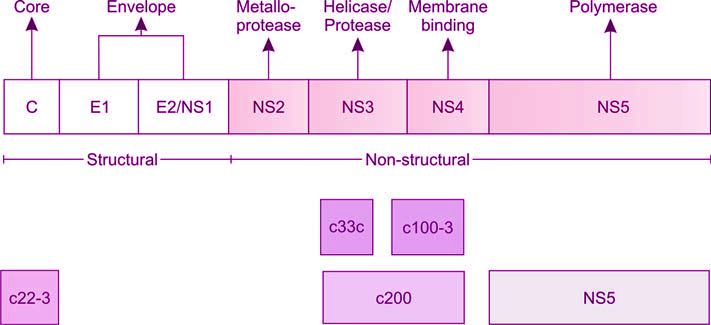
The earlier tests (first generation ELISA) used recombinant proteins complementary to the NS4 region (c100-3) of the HCV genome. Second generation ELISA incorporated recombinant or synthetic antigens from NS4 as well as NS3 regions (c100-3, c22-3, c33c) of the genome, resulting in improvement in sensitivity and specificity. The third generation ELISA, in addition to NS3 and NS4, also includes antigens from NS5 region (Figure 1197.3).
Human Immunodeficiency Virus
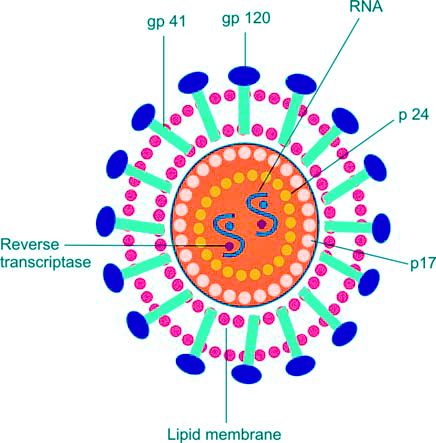
Human immunodeficiency virus or HIV is a RNA retrovirus (Figure 1197.4), which causes slowly progressive immunodeficiency in infected persons. The cells most susceptible to HIV infection are CD4+ T lymphocytes. Destruction of CD4+ T lymphocytes results in slowly progressive impairment of the immune system. The infected individual becomes susceptible to a range of opportunistic infections and malignancies. The most advanced stage of HIV disease is acquired immune deficiency syndrome or AIDS.
According to the estimates of India’s National AIDS Control Organization (NACO), adult prevalence of HIV infection is 0.7% with approximately 4 million HIV infections, 90% of which are in the age group of 15-45 years.
Following HIV infection, viremia becomes detectable after a few days and lasts for several weeks. Anti-HIV antibodies appear 6-12 weeks after infection (called as seroconversion). Principle of ELISA test for detection of anti-HIV antibodies is shown in Figure 1197.5. Window period is the period between the onset of HIV infection and appearance of detectable anti-HIV antibodies in serum; it is the infectious but seronegative period (i.e. the test for anti-HIV antibodies is yet to become positive). Transfusion of donor blood collected during the window period will transmit HIV to the recipient. According to American Association of Blood Bank standards, HIV screening tests necessary for whole blood donors are (i) Anti-HIV antibodies (HIV-1 and HIV-2) by ELISA, and (ii) HIV-1 nucleic acid amplification testing (NAT). NAT testing is based on polymerase chain reaction.
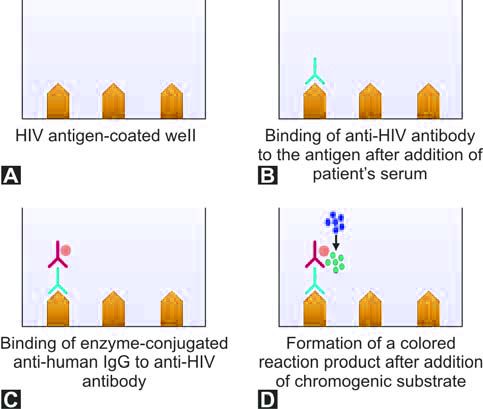
BACTERIA
Treponema pallidum
Blood transfusion can transmit Treponema pallidum, the causative agent for syphilis. It is a spirochete that can be visualized by dark ground illumination (Figure 1197.6). T. pallidum is destroyed by storage of blood at 2-8°C for 48-72 hours. However, fresh blood or platelet concentrates can transmit these organisms. Transfusion-transmission of syphilis is, therefore, rare. The main value of testing donor blood for T pallidum is to identify and exclude donors with high-risk behavior and thus who are at risk of having sexually transmitted infections.

Screening of donor blood for antibody to T. pallidum is usually carried out by rapid plasma reagin test or Venereal Disease Research Laboratory (VDRL) test. In VDRL test that is a non-specific test, donor serum (heated to 56°C to inactivate the complement) is mixed with cardiolipinlecithin-cholesterol antigen. If flocculation is observed, the test is reported as reactive (Figure 1197.7).
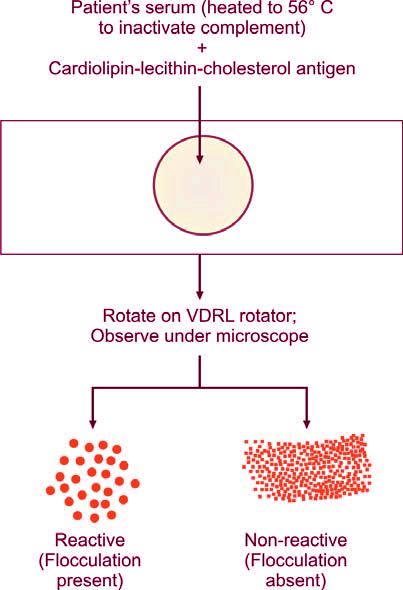
PARASITES
Plasmodium Species
Malaria parasite can be transmitted through all blood components. For detection of malaria parasite, commonly blood smears are examined by light microscopy. However, the test is positive only when parasites are > 100/μl in blood.
- Comment
- Posted by Dayyal Dg.