Microscopic Examination of Feces
Discover the significance of stool examination in detecting gastrointestinal diseases and parasitic infections. Learn proper collection methods and testing procedures for stool exams.

Microscopic examination of feces is a crucial diagnostic tool in identifying various infections and conditions related to the gastrointestinal tract. This process involves studying small samples of fecal material under a microscope to detect the presence of parasites, eggs, larvae, and other microorganisms.
Microscopic examinations done on fecal sample are shown in Flowchart 1.
- Microscopic examination of feces
- Direct wet mount
- For Eggs/larvae of helminths and trophozoites/cysts of protozoa
- If negative, concentration technique
- For Eggs/larvae of helminths and trophozoites/cysts of protozoa
- Special stains
- (A) Trichrome stain for indentification of trophozoites and cysts
(B) AFB stain for oocysts of Cryptosporidium Cyclospora, and Isospora
(C) Wright's stain for white blood cells
- (A) Trichrome stain for indentification of trophozoites and cysts
- Cellophane technique
- For eggs of Enterobius vermicularis
- Direct wet mount
Collection of Specimen for Parasites
Collect a small amount of stool (at least 4 ml or 4 cm³) in a clean container with a tight lid, like a tin box, plastic box, glass jar, or waxed cardboard box. Take 20-40 grams of formed stool or 5-6 tablespoons of watery stool. Make sure it's not mixed with urine, water, soil, or menstrual blood. Trophozoites of Entameba histolytica degrade quickly, so bring the sample to the lab immediately. Parasites are best seen in warm, fresh stools, so examine them within an hour of collection. If there's a delay, refrigerate the sample. Use a fixative with 10% formalin or polyvinyl alcohol if transporting to another lab.
Getting one negative result for parasites doesn't mean there's no infection. To be thorough, take three samples on separate days, with a 3-day gap.
For accurate results, the patient should avoid oily laxatives, antidiarrheal meds, bismuth, tetracycline antibiotics, or antacids for a week before the stool exam. Also, no barium swallow examination.
In the lab, check the stool for consistency (watery, loose, soft, or formed), color, odor, and the presence of blood, mucus, adult worms, or tapeworm segments. See Figure 1 for details.
Color/Appearance of Fecal Specimens
- Brown: Normal
- Black: Bleeding in upper gastrointestinal tract (proximal to cecum), Drugs (iron salts, bismuth salts, charcoal)
- Red: Bleeeding in large intestine, undigested tomatoes or beets
- Clay-colored (gray-white): Biliary obstruction
- Silvery: Carcinoma of ampulla of Vater
- Watery: Certain strains of Escherichia coli, Rotavirus enteritis, cryptosporidiosis
- Rice water: Cholera
- Unformed with blood and mucus: Amebiasis, inflammatory bowel disease
- Unformed with blood, mucus, and pus: Bacillary dysentery
- Unformed, frothy, foul smelling, which float on water: Steatorrhea.
Preparation of Slides
In the lab, we make saline and iodine wet mounts of the sample (see Figure 2).
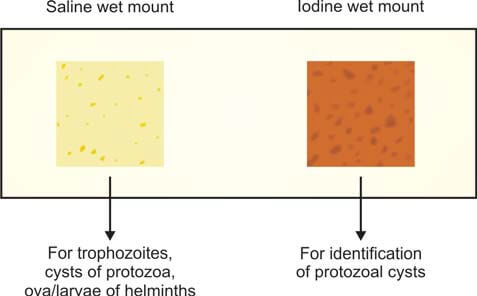
Here's how it's done: On a glass slide, put a drop of normal saline on one end and a drop of Lugol iodine solution on the other. Take a small bit of feces (about the size of a match-head) and mix it with a drop each of saline and iodine, using a wire loop. Cover each preparation with a cover slip. If there's blood or mucus in the specimen, include that part for examination (trophozoites are often found in mucus). If the stools are liquid, pick the surface portion for examination.
The saline wet mount helps show eggs, larvae of helminths, and trophozoites and cysts of protozoa. It can also detect red and white cells. Iodine stains glycogen and nuclei of cysts. The iodine wet mount is handy for identifying protozoal cysts. Trophozoites stop moving in iodine mounts. If the stool is liquid or diarrheal, you can check it directly without adding saline.
Concentration Procedure
If there are only a few parasites, concentrating the fecal specimen helps. But, it's a trade-off—while it makes it harder to detect amebic trophozoites, it's necessary when wet mount examination doesn't find anything, and there's a suspicion of a parasitic infection. This method is useful for spotting ova, cysts, and larvae of parasites.
There are two main types of concentration techniques:
- Sedimentation techniques: Ova and cysts settle at the bottom, but too much debris can make it tricky to find parasites. For example, there's the Formolethyl acetate sedimentation procedure.
- Floatation techniques: Ova and cysts float on the surface, but not all of them float in this method. Examples include the Saturated salt floatation technique and zinc sulphate concentration technique.
The commonly used sedimentation method is the Formol-ethyl acetate concentration method because:
- It can detect eggs, larvae of almost all helminths, and cysts of protozoa.
- It preserves their shape well.
- It's rapid.
- There's minimal risk of infection for lab workers because formalin kills pathogens.
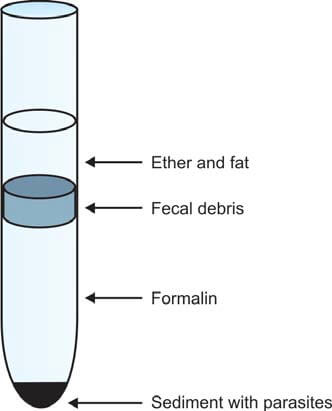
Here's how it works: Make a fecal suspension in 10% formalin (10 ml formalin + 1 gram feces). Pass it through a gauze filter until you get 7 ml of filtered material. Add ethyl acetate (3 ml), then centrifuge the mixture for 1 minute. Eggs, larvae, and cysts settle at the bottom (see Figure 3). Remove the layers above the deposit—formalin, fecal debris, and ether. Loosen the debris, pour off the supernatant, and place a drop of sediment on each end of a glass slide. Stain one drop with iodine, cover slips, and examine it under the microscope.
Classification of Intestinal Parasites of Humans
Humans' intestinal parasites fall into two main groups: protozoa and metazoa (helminths) (see Flowchart 2).
- Intestinal parasites
- Protozoa
- Amebae: Entamoeba histolytica
Flagellates: Gardia lamblia
Ciliates: Balantidium coli
Coccidia: Isospora belli, Cryptosporidium parvum, Cyclospora cayetanensis
Microsporidia
- Amebae: Entamoeba histolytica
- Helminths
- Nemathelminthes
- Ascaris lumbricoides
Enterobius vermicularis
Ancylostoma duodenale
Necator americanus
Strongyloides stercoralis
Trichuris trichiura
- Ascaris lumbricoides
- Platyhelminthes
- Trematodes
- Fasciolopsis buski
- Cestodes
- Taenia saginata
Taenia solium
Hymenolepis nana
Diphyllobothrium latum
- Taenia saginata
- Trematodes
- Nemathelminthes
- Protozoa
Summary
Microscopic examination of feces is a crucial diagnostic tool in identifying various infections and conditions related to the gastrointestinal tract. This process involves studying small samples of fecal material under a microscope to detect the presence of parasites, eggs, larvae, and other microorganisms.
Procedure
- Sample Collection: A small amount of stool is collected and prepared for examination. The sample should be free from contamination with urine, water, or soil.
- Wet Mount Preparation: The sample is mixed with normal saline or iodine solution on a glass slide. This helps in observing live organisms and provides information on their motility and characteristics.
- Concentration Techniques: In some cases, concentration methods are used to increase the chances of detecting parasites. Sedimentation and floatation techniques are common, helping to separate parasites from fecal debris.
- Microscopic Observation: The prepared slides are examined under a microscope. The pathologist looks for parasites, cysts, eggs, larvae, and other relevant structures. The examination may also include assessing the color, consistency, and presence of blood or mucus.
Significance
- Parasitic Infections: Microscopic examination helps identify a wide range of parasitic infections, including protozoa and helminths.
- Disease Diagnosis: It aids in diagnosing conditions such as amoebiasis, giardiasis, and various helminth infections.
- Treatment Monitoring: Monitoring the presence of parasites is essential to track the effectiveness of treatment and ensure the elimination of the infection.
In Brief
Microscopic examination of feces is a valuable tool in the hands of pathologists to diagnose and monitor gastrointestinal infections. It allows for a detailed analysis of the fecal sample, aiding in the timely and accurate identification of parasites and other microscopic elements that may indicate an underlying health issue.
The information on this page is peer reviewed by a qualified editorial review board member. Learn more about us and our editorial process.
Last reviewed on .
Article history
- Latest version
Reference(s)
- American Gastroenterological Association. AGA technical review on the evaluation and management of chronic diarrhea. Gastroenterology 1999;116: 1464-86.
- American Gastroenterological Association Medical Position Statement: Guidelines for the evaluation and management of chronic diarrhoea. Gastroenterology 1999;116:1461-3.
- Haque R, Huston CD, Hughes M, Houpt E, Petri, WA Jr. Amebiasis. New Engl J Med 2003;348:1565:73.
- Kucik CJ, Martin GL, Sortor BV. Common intestinal parasites. Am Fam Physician 2004;69:1161-8.
Cite this page:
- Comment
- Posted by Dayyal Dungrela