Microscopic Examination of Blood for Demonstration of Microfilaria
Since some species exhibit periodicity (i.e. circulation of microfilariae in increased numbers at certain times of the day), blood should be collected at the correct time to improve the chances of detection. For Wuchereria bancrofti and Brugia malayi showing nocturnal periodicity, blood should be collected at night between 10 p.m. to 4 a.m. Microfilariae are present in greater numbers in capillary blood than in venous blood; therefore, skin puncture is preferred. Usually microfilariae are scanty in peripheral blood so that concentration techniques may be necessary for their demonstration.
Following microscopic methods can be used for detection of microfilariae in peripheral blood:
- Thick blood smear
- Concentration techniques: mem-brane filtration, microhematocrit centrifugation, lysed venous blood technique, lysed capillary blood technique.
Thick blood smear
A thick blood smear is spread from 20 μl of capillary blood on a glass slide, air-dried, and stained with a Romanowsky stain. If microfilariae are not detected in thick smears prepared from capillary blood collected at the appropriate time, and if clinical suspicion is strong, concentration techniques are employed. This is because circulating microfilariae are often scanty and sensitivity of microfilarial detection increases when volume of blood sampled is increased.
Concentration techniques
Membrane filtration
This is a sensitive method but is expensive for routine use in endemic areas. Anticoagulated venous blood (10 ml) is passed through a polycarbonate membrane filter of 3 μm or 5 μm pore size. Following this 10 ml of methylene blue saline solution is passed through the filter for staining the microfilariae. Microfilariae are trapped and retained on the filter, which is placed on a glass slide and examined under the microscope.
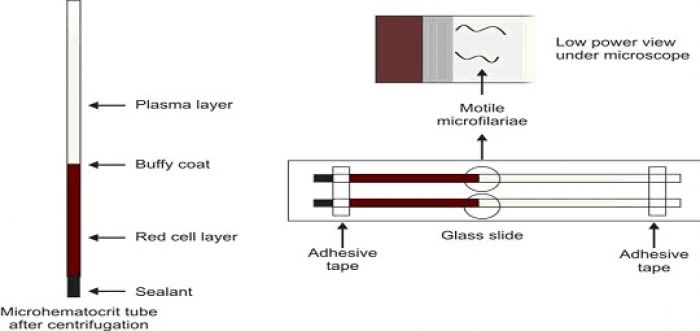
Microhematocrit tube or capillary tube method
Two heparinised capillary tubes are filled with blood from skin punctures (or two plain capillary tubes are filled with anticoagulated venous blood). After sealing the dry ends with a suitable sealant, tubes are centrifuged in a microhematocrit centrifuge for about 5 minutes. The capillary tubes are placed on a glass slide and fixed with adhesive tape. Plasma just above the buffy coat layer is examined for motile microfilariae under the microscope.
Lysed venous blood method
10 ml of venous blood is lysed by saponin-saline solution. The hemolysate is centrifuged, supernatant is discarded, and the sediment is placed on glass slide. After adding a drop of methylene blue solution, a coverslip is placed, and the preparation is examined under the microscope for microfilariae.
Lysed capillary blood method
0.1 ml of blood obtained by skin puncture is added to 1 ml of saponin-saline solution to cause lysis of red cells. After centrifugation, supernatant is discarded and sediment is placed on a glass slide. A drop of methylene blue solution is added and a coverslip is placed over it. The entire preparation is examined under the microscope for motile microfilariae.
Morphology of Microfilariae on Romanowsky-stained Blood Smears Wuchereria bancrofti
Microfilariae measure about 300 μ in length and 8 μ in breadth. They have a hyaline sheath, which stains pink. There are distinct nuclei in the central axis of the body. Nuclei are not present in the tip of the tail. Cephalic space (present at the anterior end) is as long as it is broad. Tip of the tail is bent backwards and body curves are few.
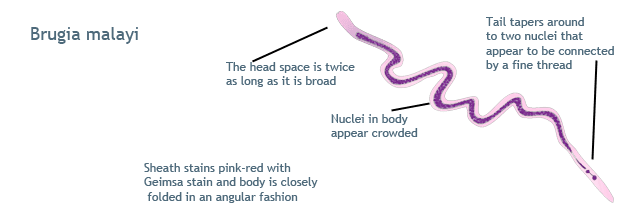
Brugia malayi: These measure about 230 μ × 6 μ in size. Sheath stains dark pink in color. The nuclei are crowded in the body, are blurred in outline, and the tip of the tail shows two distinct nuclei. Cephalic space is twice as long as it is broad. Instead of smooth curves to the body, there are kinks.
- Comment
- Posted by Dayyal Dg.