Medically reviewed and approved by a board-certified member
FLOW CYTOMETRY
 Flow cytometry is a procedure used for measuring multiple cellular and fluorescent properties of cells when they flow as a single cell suspension through a laser beam by a specialized instrument called as a flow cytometer. Flow cytometry can analyze numerous cells in a short time and multiple parameters of a single cell can be analyzed simultaneously. From the measured parameters, specific cell populations are defined. Cells or particles with size 0.2-150 μm are suitable for flow cytometer analysis.
Flow cytometry is a procedure used for measuring multiple cellular and fluorescent properties of cells when they flow as a single cell suspension through a laser beam by a specialized instrument called as a flow cytometer. Flow cytometry can analyze numerous cells in a short time and multiple parameters of a single cell can be analyzed simultaneously. From the measured parameters, specific cell populations are defined. Cells or particles with size 0.2-150 μm are suitable for flow cytometer analysis.Flow cytometry can provide following information about a cell (Box 807.1):
- Cell size (forward scatter)
- Internal complexity or granularity (side scatter)
- Relative fluorescence intensity.
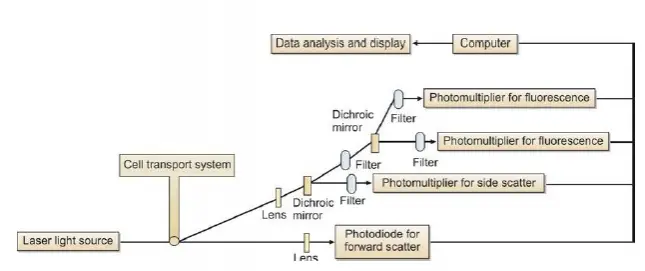
A flow cytometer consists of three main components or systems: fluidics, optics, and electronics.
(1) Fluidics: The function of the fluidics system is to transport cells in a stream to the laser beam for interrogation. Cells (fluorescence-tagged) are introduced into the cytometer (injected into the sheath fluid within the flow chamber) and made to flow in a single file past a laser (light amplification by stimulated emission of radiation) beam. The stream transporting the cells should be positioned in the center of the laser beam. The portion of the fluid stream where the cells are located is called as the sample core. Only a single cell or particle should pass through the laser beam at one time. Flow cytometers use the principle of hydrodynamic focusing (process of centering the sample core within the sheath fluid) for presenting cells to the laser.
(2) Optics: This system consists of lasers for illumination of cells in the sample, and filters to direct the generated light signals to the appropriate detectors.
The light source used in most flow cytometers is laser.
The laser most commonly used in flow cytometry is Argon-ion laser. The light signals are generated when the laser beam strikes the cell, which are then collected by appropriately positioned lenses. A system of optical mirrors and filters then directs the specified wavelengths of light to the designated detectors. Two types of light signals are generated when the laser beam strikes cells tagged with fluorescent molecules: fluorescence and light scatter. The cells tagged with fluorescence emit a momentary pulse of fluorescence; in addition, two types of light scatter are measured: forward scatter (proportional to cell diameter) and side scatter (proportional to granularity of cell).
(3) Electronics: The optical signals (photons) are converted to corresponding electronic signals (electrons) by the photodetectors (photodiodes and photomultiplier tubes). The electronic signal produced is proportional to the amount of light striking a cell. The electric current travels to the amplifier and is converted to a voltage pulse. The voltage pulse is assigned a digital value representing a channel by the Analog-to Digital Converter (ADC). The channel number is then transferred to the computer which displays it to the appropriate position on the data plot.
Further Reading:
- Comment
- Posted by Dayyal Dg.
Tags:
End of the article